PsychoGenics leverages rigorous, well-validated testing methods to conduct the highest-quality preclinical depression studies for your innovative treatment. Rooted in a deep understanding of depression and its underlying mechanisms, PsychoGenics is equipped with a diverse range of preclinical tests designed to catalyze your research breakthroughs.
Validated Tests for Depression
PsychoGenics’ comprehensive battery of behavioral assessments offers a thorough preclinical evaluation of depressive-like behaviors and gauges the effectiveness of prospective treatments:
Chronic Social Defeat Stress (CSDS) is a behavioral assay that reflects the wide range of responses individuals have to physical and psychological stressors. This method is crucial in preclinical depression studies, capturing outcomes that vary from the onset of major depressive disorder and post-traumatic stress disorder to resilience against these states. CSDS is particularly valuable for its high construct, face, discriminative, and predictive validity in modeling anxiety disorders, such as PTSD. This approach is integral to developing validated depression disease models and contributes significantly to the understanding of stress-related disorders in early-stage depression research.
Control and Defeat Phenotype
Within the framework of Chronic Social Defeat Stress research, the Control and Defeat Phenotype observed at PsychoGenics shows that susceptible mice demonstrate a marked reduction in social interaction when introduced to a novel mouse. Specifically, during the social interaction test, these mice tend to spend more time in the corners of the arena, avoiding the interaction zone where the novel mouse is placed. This behavior is crucial for our depression studies and early-stage depression research, as it contributes to the development of validated depression disease models by illustrating the social avoidance seen in some stress responses.
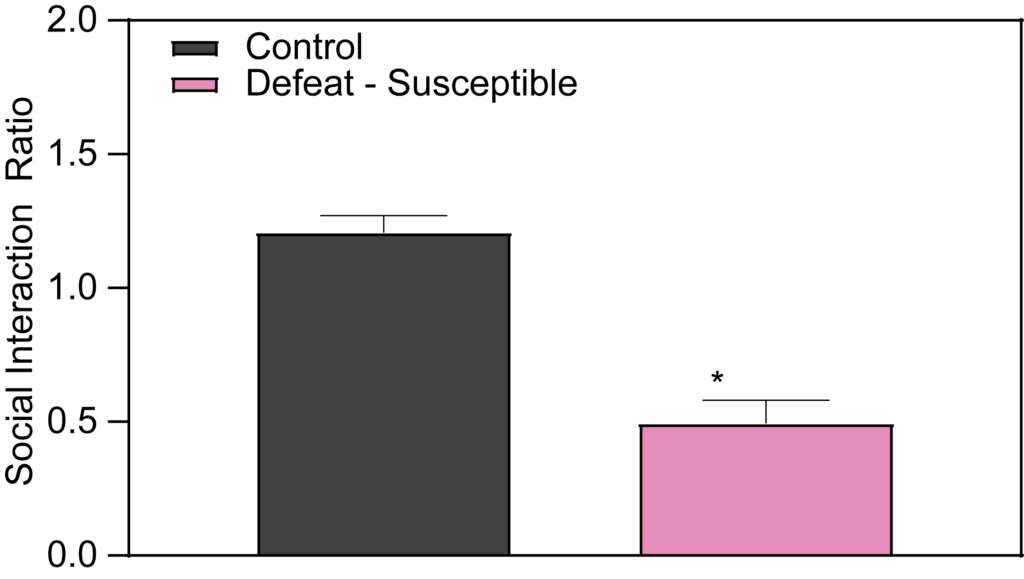
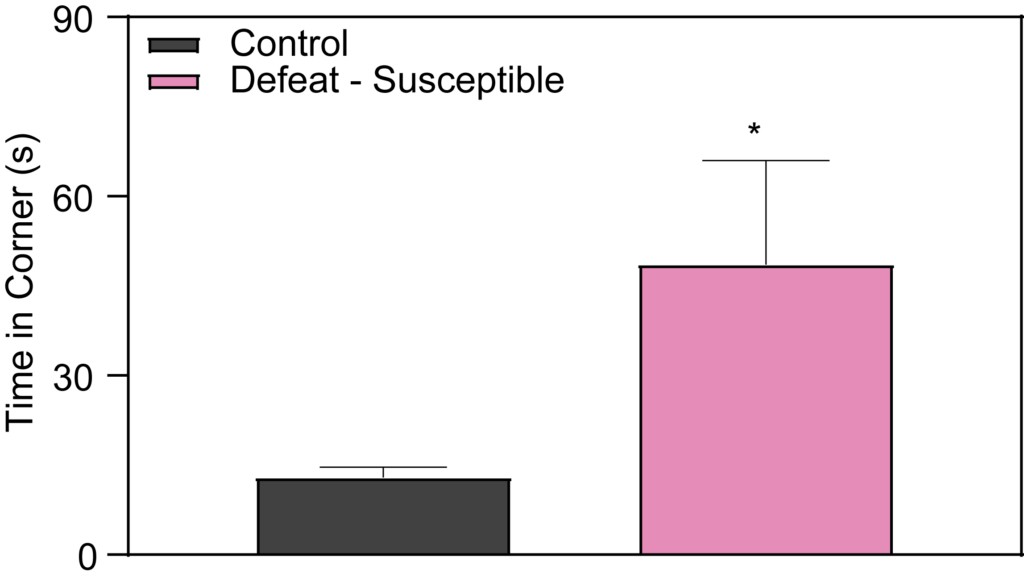
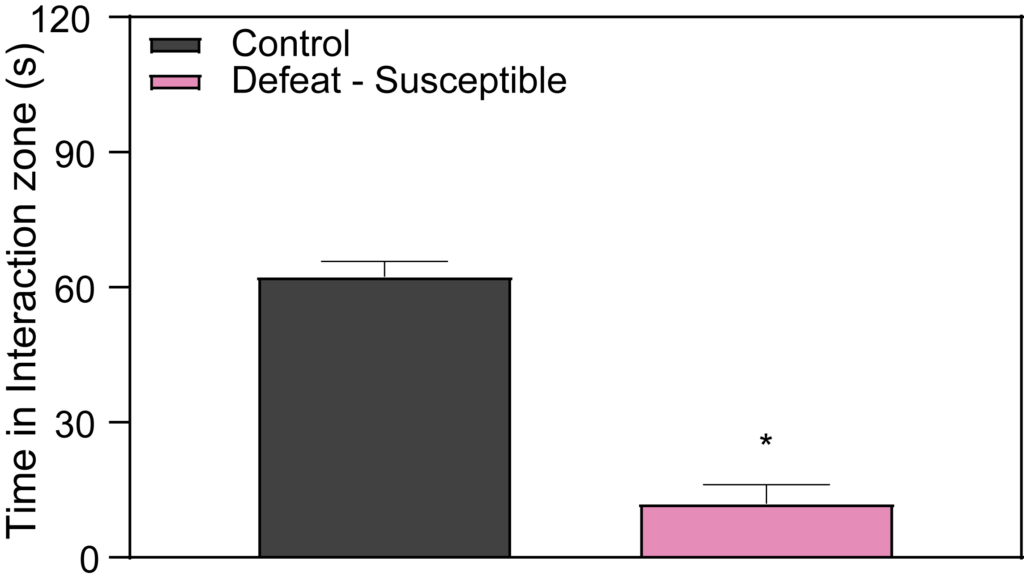
Effects of Imipramine and Ketamine on Social Interaction in Defeated Mice
Our studies at PsychoGenics also delve into the effects of Imipramine and Ketamine on social interaction in defeated mice. The results indicate that acute administration of Imipramine does not demonstrate efficacy in this model. However, when Imipramine is administered chronically (over 2 weeks), it significantly attenuates the social deficits observed in defeated mice. Conversely, an acute injection of Ketamine administered 24 hours prior to testing has been effective in attenuating social deficits in defeated mice. These findings are instrumental for our depression studies and early-stage depression research, highlighting potential pharmacological interventions for stress-induced social avoidance.
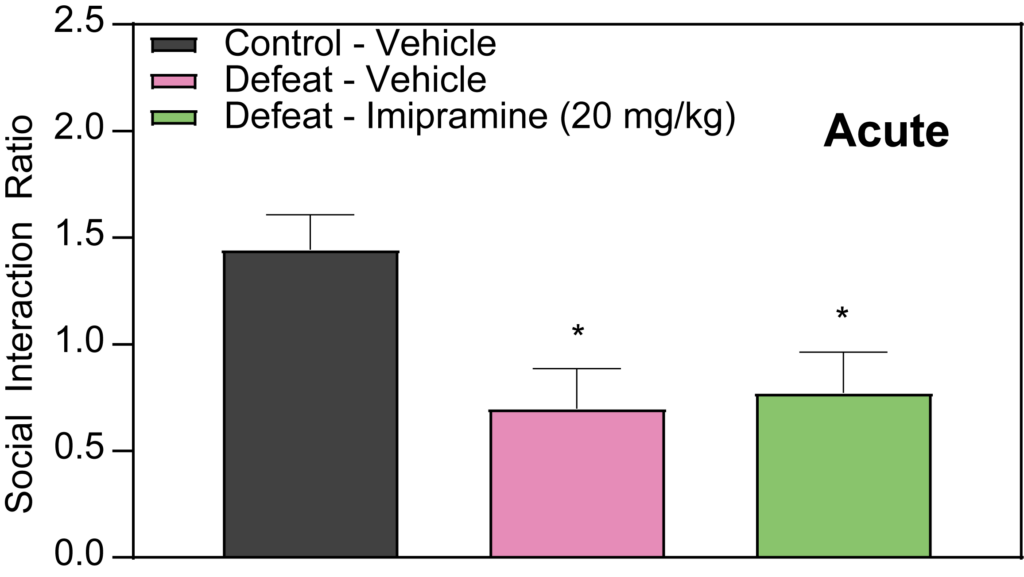
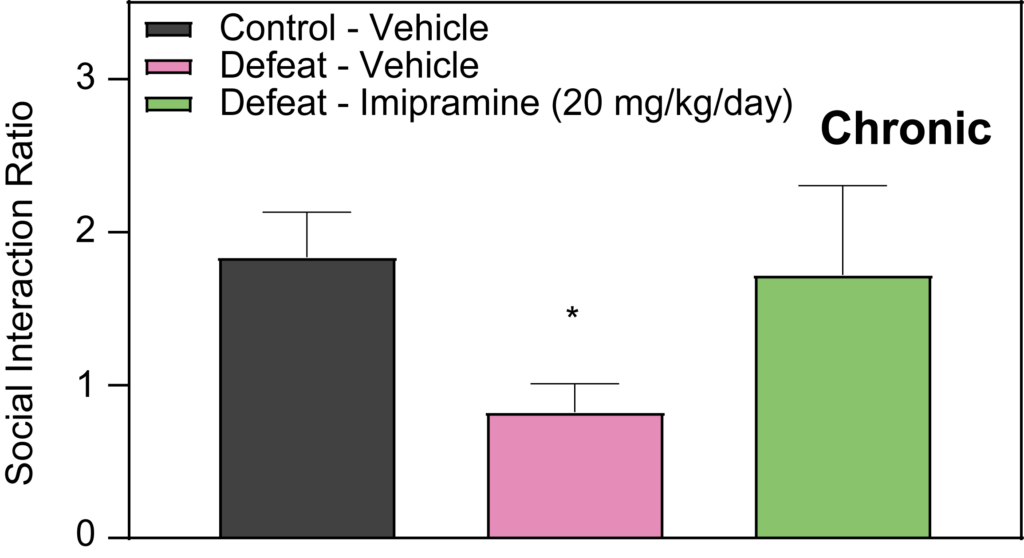
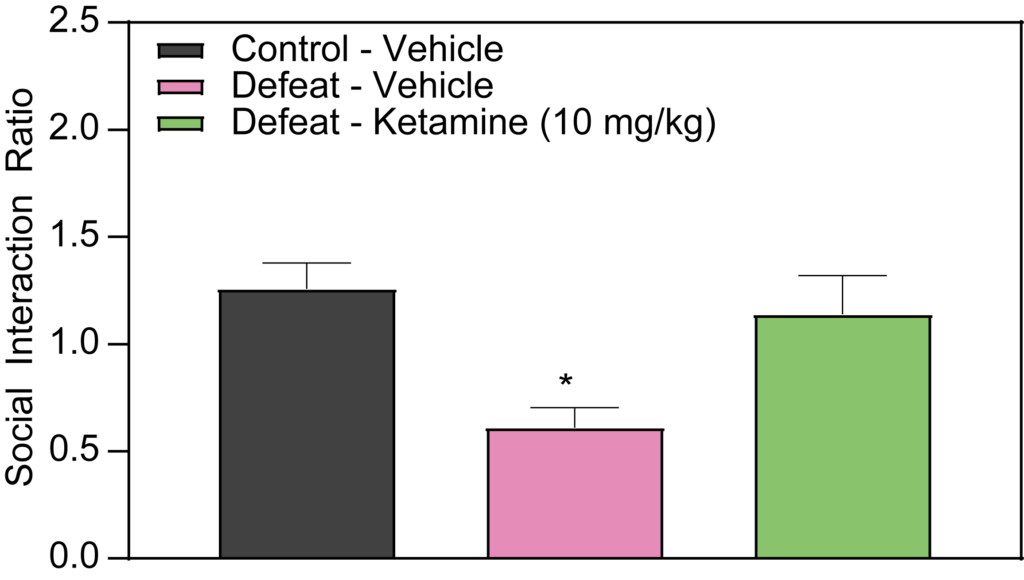
Differential Reinforcement of Low Rate 72-second schedule (DRL72s) is an operant test used in preclinical depression studies for behavioral suppression, requiring a pause in response for 72 seconds. This test is key in developing validated depression disease models because it measures how antidepressants, like tricyclics, MAOIs, and SSRIs, can decrease response rates while increasing reinforcement rates. Non-antidepressants, such as psychostimulants, show an opposite effect. These findings support early-stage depression research by differentiating the drug responses, crucial for confirming the predictive validity of antidepressant treatments.
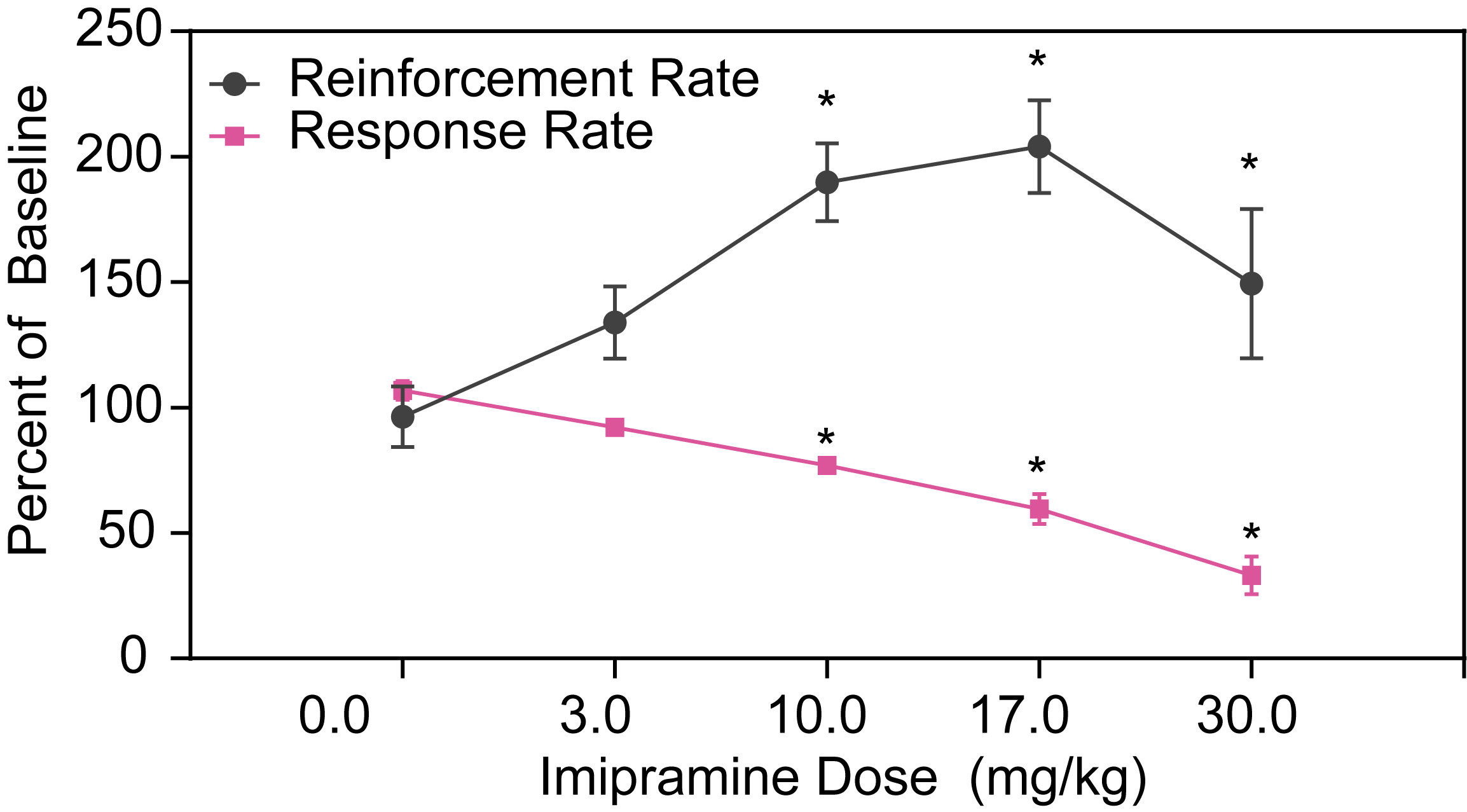
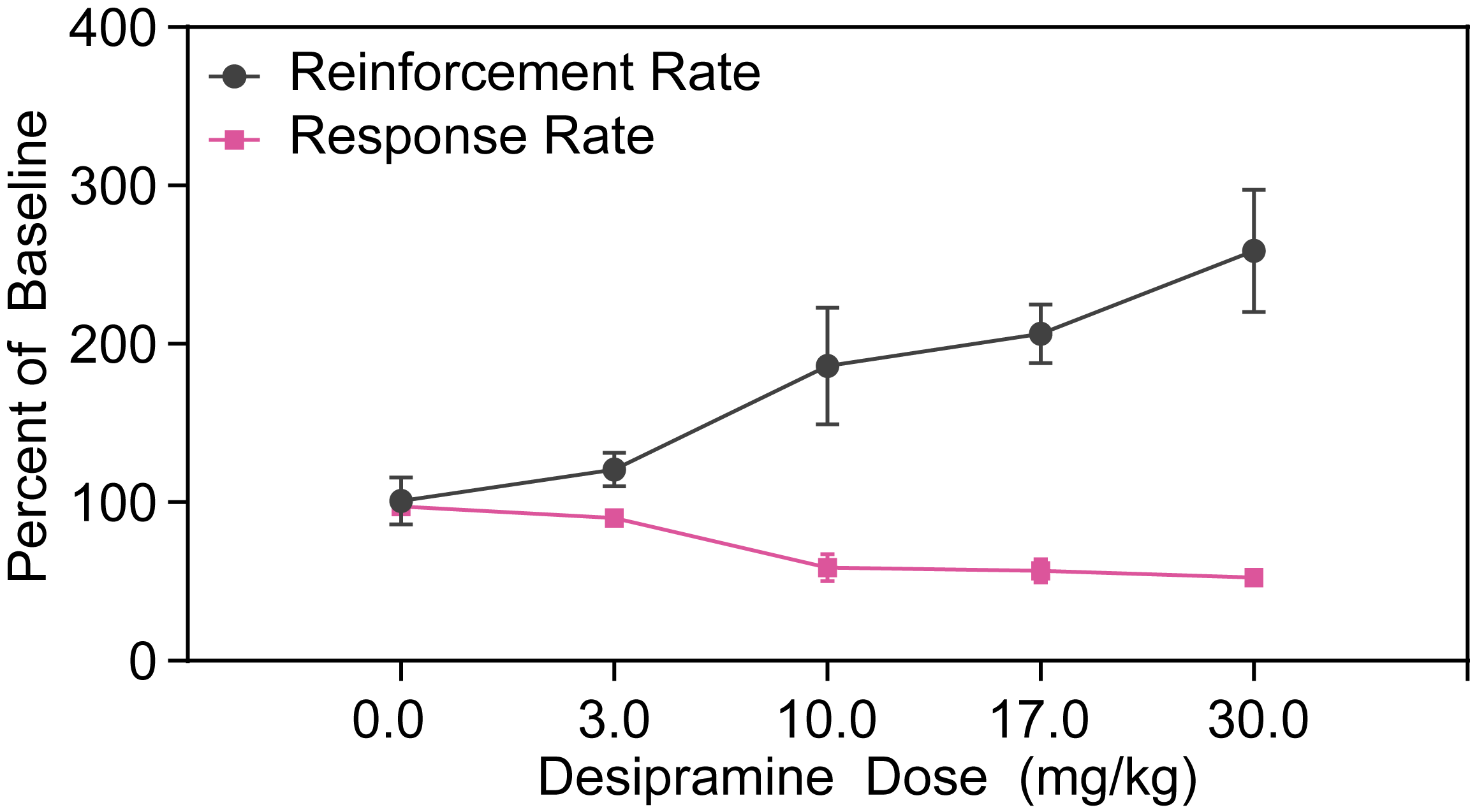
Effects of Tricyclic Antidepressants in DRL72 in Rats
In DRL72s tests focused on rats, tricyclic antidepressants like Imipramine and Desipramine have been shown to increase reinforcement rates while decreasing response rate. These effects are valuable for validating depression disease models in preclinical depression studies, contributing to the understanding of antidepressant efficacy in early-stage depression research.
The forced swim test is a widely recognized tool in preclinical depression studies, where rats or mice are placed in a deep cylinder filled with tepid water, leading to a state of reduced mobility or “behavioral despair.” This immobility is consistently lessened when the animals are treated with various antidepressants, underscoring the test’s relevance in validated depression disease models. Rodent models of depression are primarily valued for their predictive validity, the objectivity of the measured responses, and their high reliability and reproducibility, all of which are critical in the progression of early-stage depression research.
Effects of SSRI and Tricyclic Antidepressants
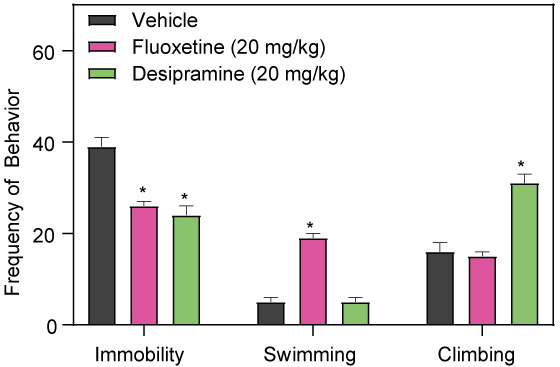
The forced swim test in rats can differentiate between SSRI and Tricyclic-like antidepressants by their differential effects on climbing and swimming behaviors following sub-chronic intraperitoneal injection.
Effects of Double and Triple Reuptake Inhibitors
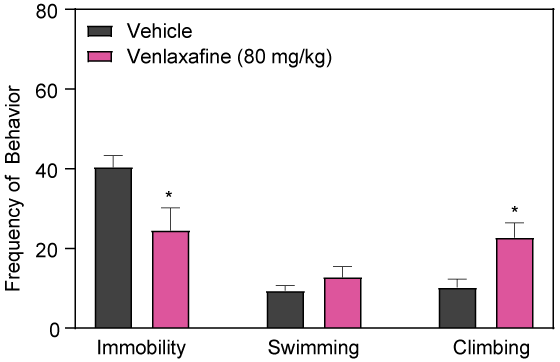
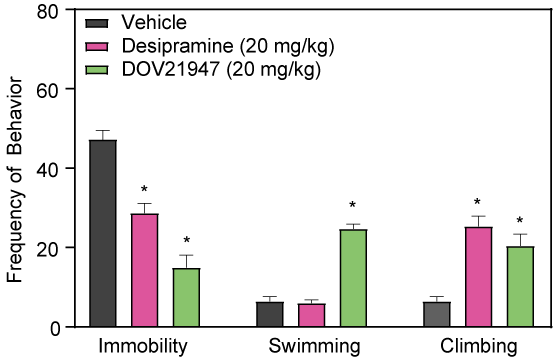
The triple uptake inhibitor DOV21947 and the double (serotonin / norepinephrine) reuptake inhibitor, venlafaxine, decrease immobility in the rat FS test following sub-chronic intraperitoneal injection.
Sertraline Decreases Time Immobile in BALB/cJ Mice
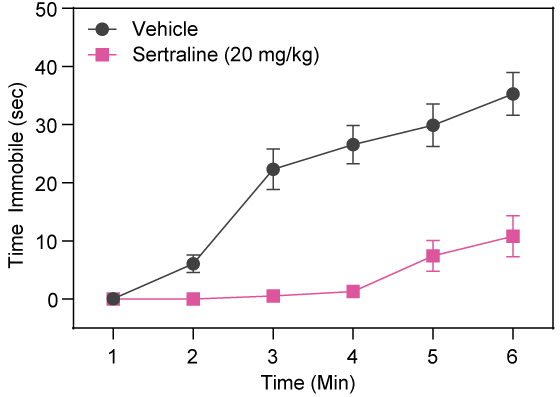
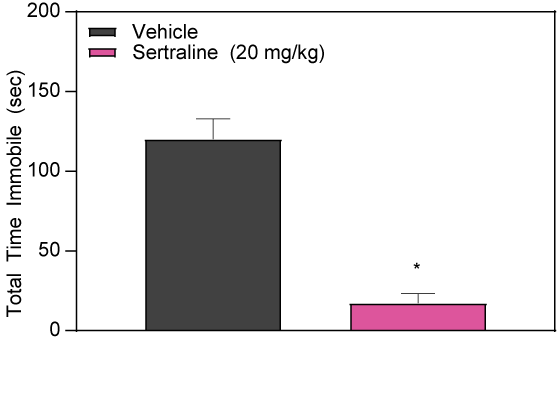
Sertraline, a Selective Serotonin Reuptake Inhibitor, decreases immobility of BALB/cJ mice in the FS test following acute intraperitoneal injection.
The marble burying assay, employed in preclinical depression studies, serves as a model for examining behaviors related to anxiety and obsessive-compulsive disorder. In this assay, mice are placed in cages with a 6cm layer of hardwood bedding and twenty evenly spaced black marbles for a duration of 30 minutes. Their activity is monitored by overhead cameras, with the distance traveled recorded and analyzed using specialized video tracking software. The primary measure is the number of marbles buried, with a marble deemed buried when two-thirds of it is submerged in the bedding.
This assay is not only instrumental for observing behavior but also for investigating the pharmacological effects of drugs through agonist or antagonist studies, providing insight into their mechanisms of action. Additionally, it offers surrogate pharmacokinetic data on a drug’s duration of action. Notably, mice pre-treated with antidepressants, including classes like tricyclics and SSRIs, demonstrate a reduced propensity to bury marbles compared to control groups. Specifically, both Desipramine and Paroxetine are shown to decrease marble-burying behavior, with Desipramine also presenting a dose-dependent reduction in locomotor activity, whereas Paroxetine does not affect locomotion.
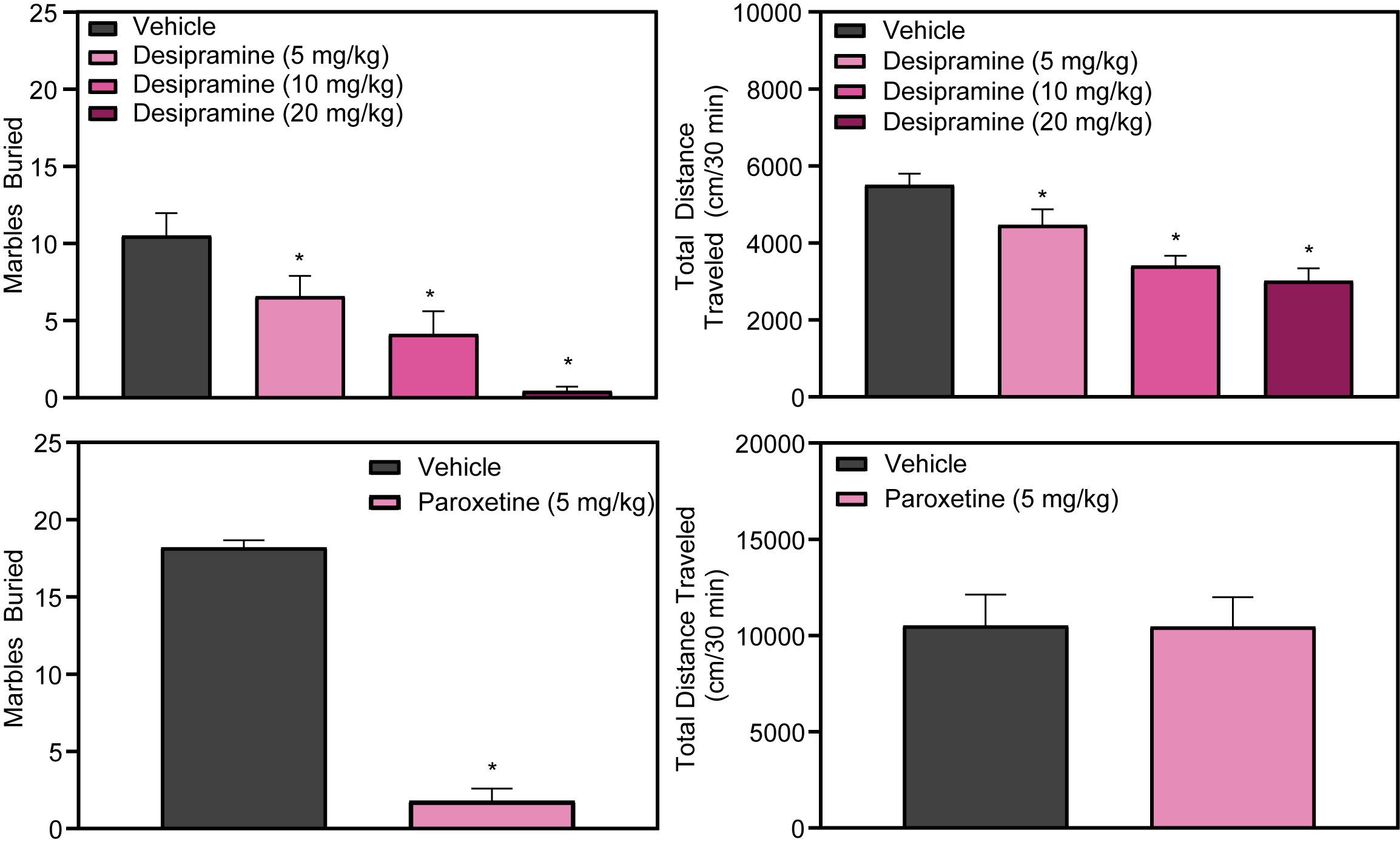
Novelty Suppressed Feeding (NSF) is an assay used in preclinical depression studies to gauge a rodent’s hesitance to eat in an unfamiliar environment. This test is pertinent for evaluating the effects of chronic antidepressant treatment. Chronic administration of Imipramine, for instance, has been shown to reduce the time it takes for rodents to begin eating, indicating decreased anxiety or aversion in the novel setting. However, it’s important to note that this does not alter their eating behavior in their familiar home cage environment. Such findings are vital for validating depression disease models and are integral to the advancement of early-stage depression research.
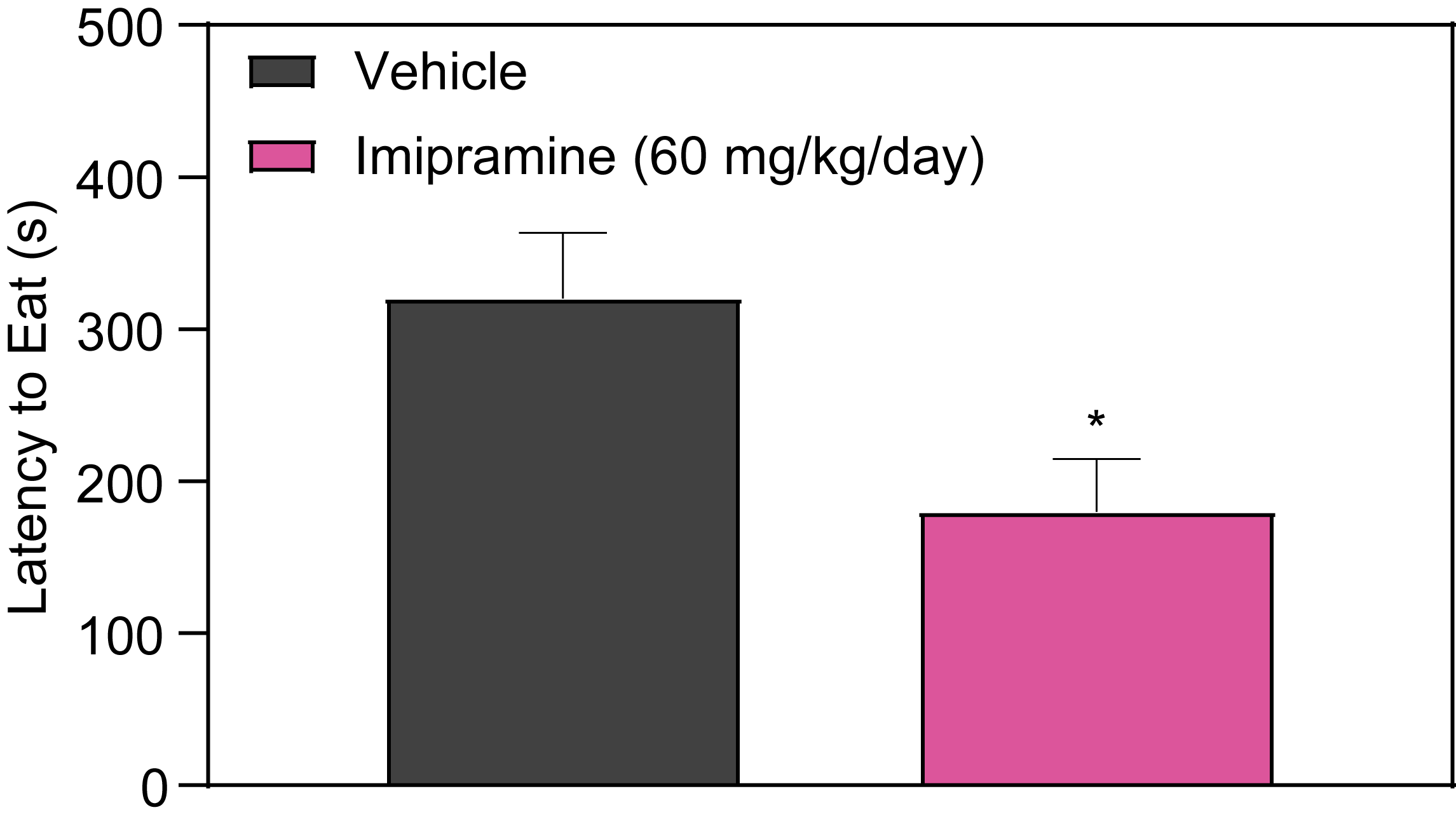
The tail suspension assay is an established method in preclinical depression studies that assesses the tendency of mice to become immobile when suspended by their tails. The duration of this immobility is typically decreased by the administration of clinically active typical and atypical antidepressants, marking the test’s utility in developing validated depression disease models. Notably, acute intraperitoneal injections of antidepressants such as Bupropion, Fluoxetine, or Desipramine have been shown to reduce the immobility of mice in the tail suspension test. These results are indicative of the potential antidepressant properties of these compounds and are critical to the progression of early-stage depression research.
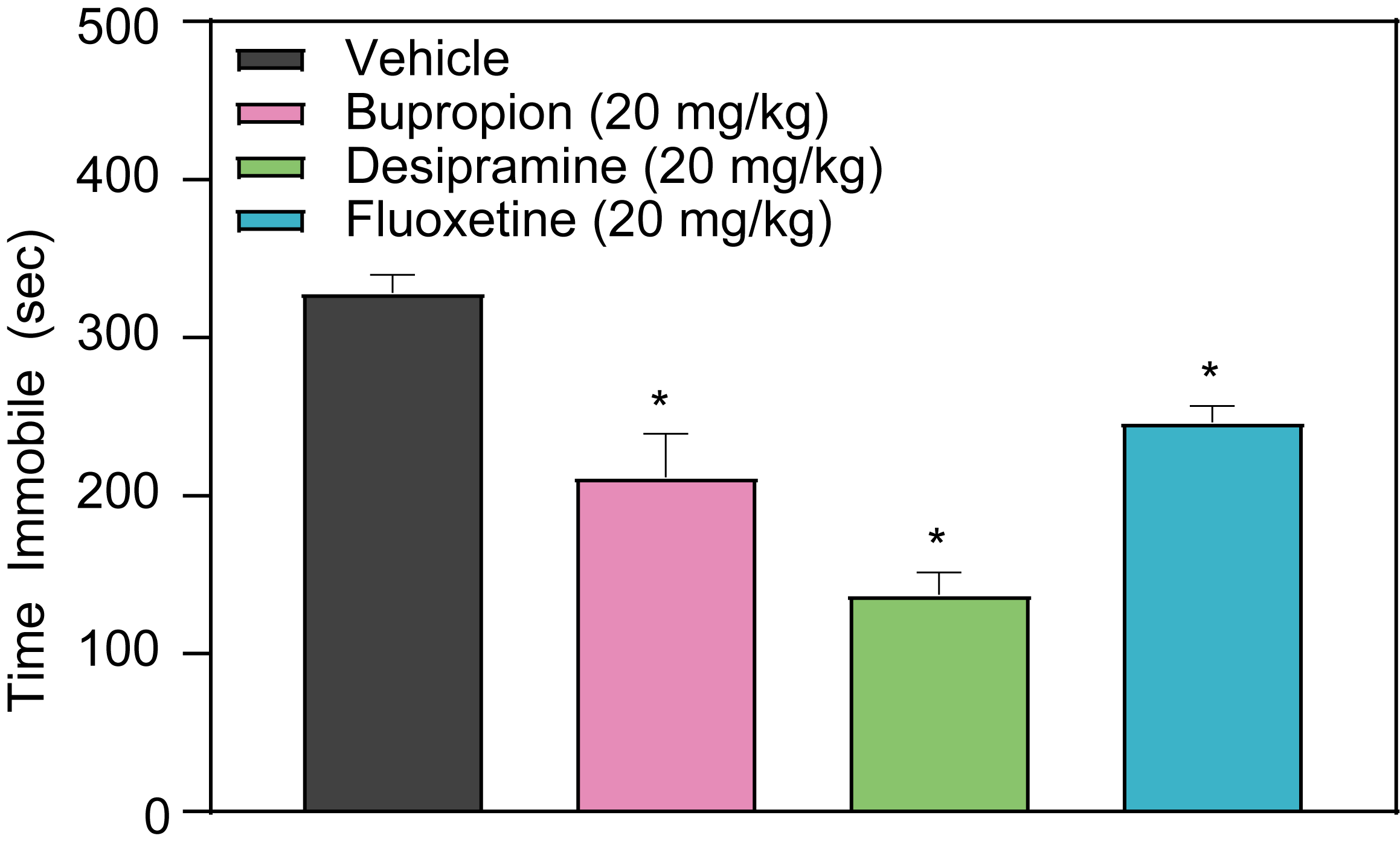
Your Trusted Partner for Preclinical Depression Research
Partner with PsychoGenics to advance your preclinical depression program and contribute to the discovery of transformative therapies for individuals battling this pervasive mental health disorder.
Explore our areas of specialization: