Autism spectrum disorders (ASDs) describe a broad category of neurodevelopmental disorders that can pose challenges to an individual’s health over a lifetime. The severity and specificity of clinical features of ASDs can vary from individual to individual, encompassing impairments in cognitive function, general social skills and abnormal sensory processing and/or responses. While the etiologies of various ASDs are not fully defined, the broad consensus holds that both environmental and genetic perturbations are contributing factors. PsychoGenics offers well-characterized and widely used genetic mouse models of several ASDs, with scientific expertise to help drive progress in your research program.
Rett Syndrome (LTP)
Rett syndrome (RTT) is a developmental X-linked genetic disorder resulting from mutations in the gene coding for the methyl CpG binding (MeCP2) protein1. The MeCP2tm1.1 Bird model lacks one copy of the MeCP2 gene2 and displays robust behavioral impairments for preclinical drug screening/testing. In addition to behavioral deficits, a significant reduction in hippocampal long-term synaptic plasticity (LTP) in female MeCP2tm1.1 Bird mice has been confirmed3, and validated by PsychoGenics.
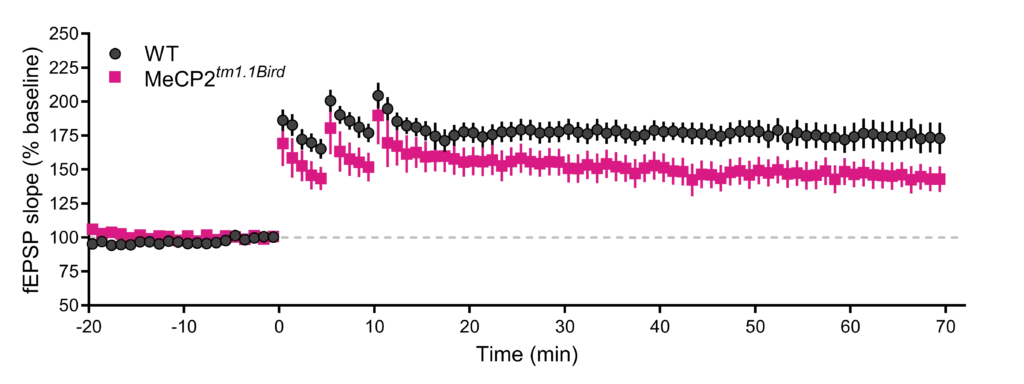

Hippocampal LTP is impaired in brain slices from Rett Syndrome mice. (Left) Time course of responses in an LTP experiment comparing extracellular field excitatory postsynaptic potentials (fEPSP) evoked in hippocampal slices (Schaffer collateral-CA1 synapse) from 6-month old female wild-type (WT) vs. MeCP2tm1.1Bird mice. LTP was induced by delivering three trains of 100 Hz high frequency stimulation, each train separated by a five-minute inter-train interval. Synaptic responses were normalized and expressed as percent (%) from baseline.(Right) Quantification of the fEPSP slope between minutes 65-70 showing reduced LTP in MeCP2tm1.1 Bird female mice (*p=0.0333, Mann-Whitney Rank Sum Test, two-tailed).
Fragile X – E/I imbalance and Tonic Currents – Deficits in Inhibition
Loss of function mutations in the Fmr1 gene (which encodes Fragile X mental retardation protein, or FMRP) causes Fragile X syndrome (FXS), the most common inheritable cause of autism and cognitive impairment4. A shift in the balance of excitatory/inhibitory neurotransmission is thought to underlie many of the behavioral phenotypes in Fragile X, including hyperactivity, sensory hypersensitivity, and seizures. FMR1 knockout male mice (FMR1_KO) display a significant reduction in tonic GABA-mediated inhibitory currents, a critical factor in modulating neuronal and network excitability5. These shifts in the balance of excitation-inhibition to favor excitation are thought to be related to the sensory disturbances in Fragile X patients.
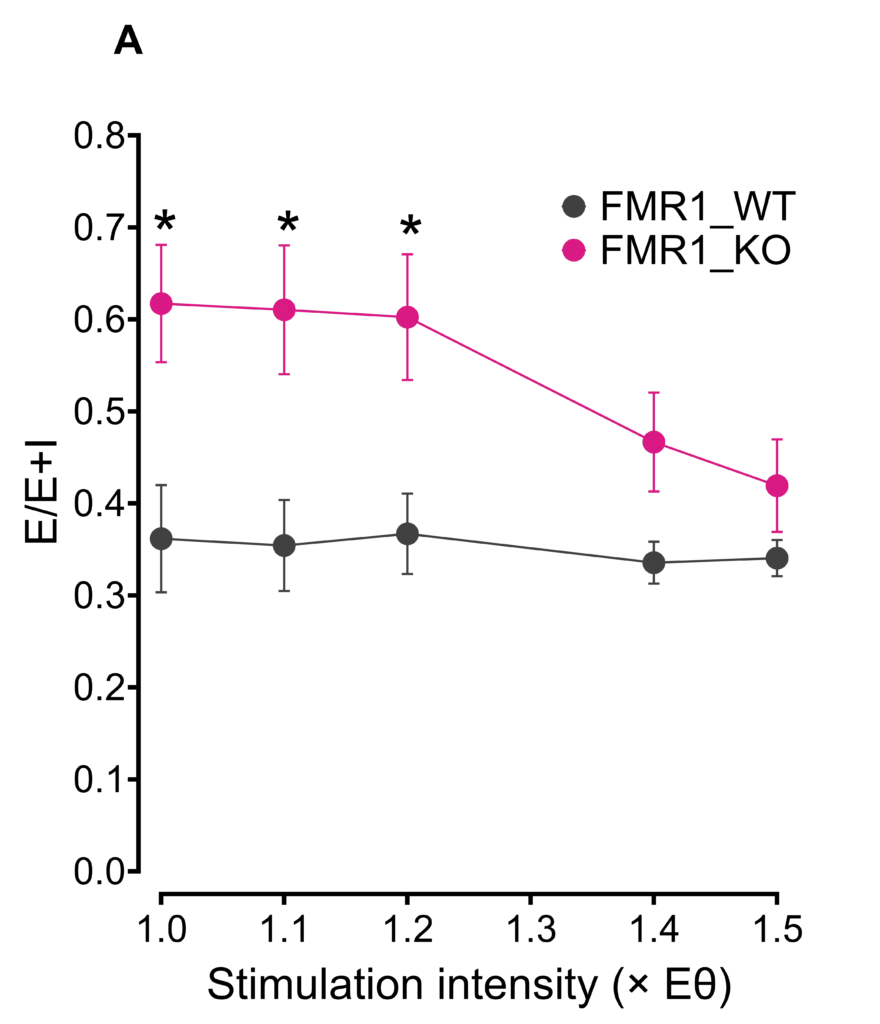
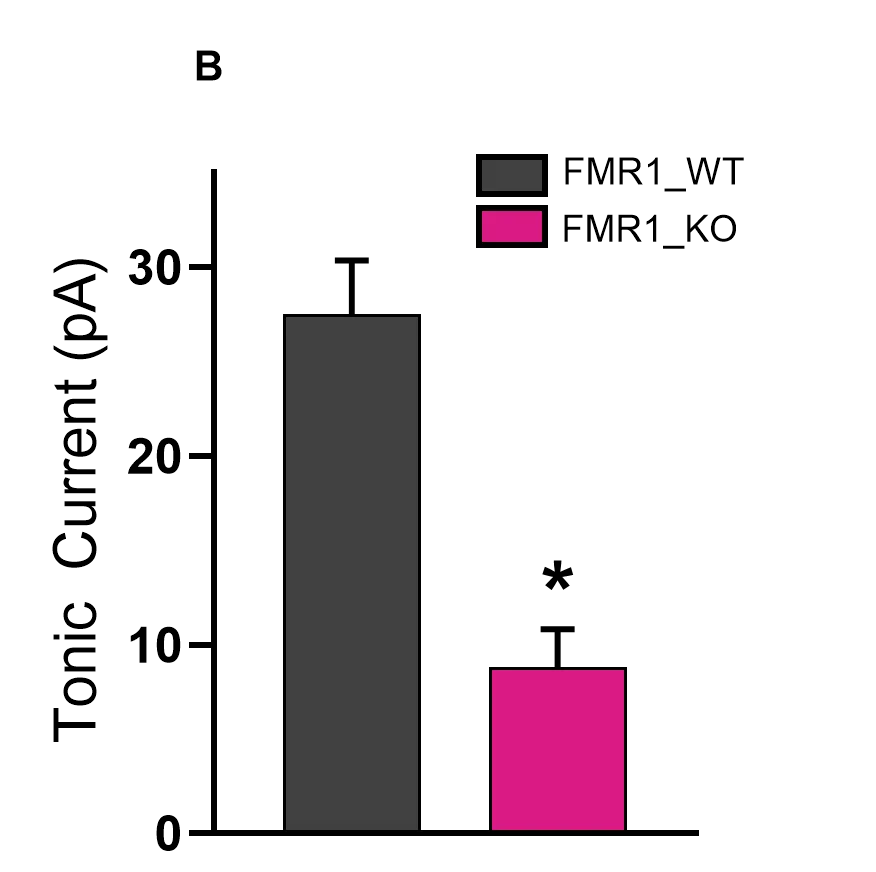
Decreases in inhibition lead to shifts in the balance between excitation/inhibition in neural circuits to favor excitation in Fragile X. (A) By measuring both excitatory and inhibitory evoked postsynaptic currents in layer II/III pyramidal neurons of the sensory cortex, we can compare the resultant ratio of excitatory and inhibitory tone in FMR1_WT and FMR1_KO mice, which clearly favors a shift to excitation in FMR1_KO. (B) Modulating tonic inhibition may represent a therapeutic strategy for several areas including autism spectrum disorders, cognitive dysfunction, and epilepsy. Inhibitory tonic current amplitudes were recorded from hippocampal dentate granule cells of hippocampal slices from 2-month-old male wild-type (FMR1_WT) and FMR1-/y (FMR1_KO) mice, after being maximized by application of the d subunit-selective agonist gaboxadol (1 mM). The degree of tonic inhibition present was then unmasked by blocking GABAA receptors with 100 mM picrotoxin demonstrating that granule cells, a critical filter to the hippocampal formation, are less inhibited in FMR1_KO mice. *p<0.05, ****p<0.0001, two-way repeated measures ANOVA, Bonferroni’s multiple comparisons test.
Shank3/F – Impaired excitatory transmission at corticostriatal synapses
Excitatory neurotransmission is impaired at corticostriatal synapses in Shank3/F mice. Clinical evidence supports involvement of striatal circuits in autism spectrum disorders (ASD) pathophysiology6. Shank family proteins are an integral part of postsynaptic density in excitatory synapses. Shank3 is enriched in striatum and its mutations have been linked to both ASD and schizophrenia7. Shank3/F mutant mice developed by Guoping Feng8 show behavioral abnormalities associated with ASD.
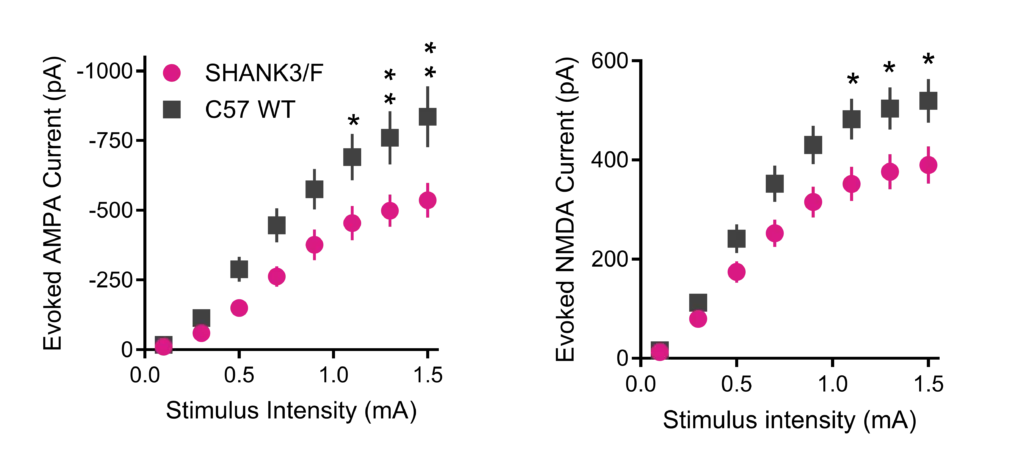
Excitatory AMPA (Left) and NMDA (Right) receptor-mediated excitatory postsynaptic currents recorded from medium spiny neurons in dorsolateral striatum evoked by stimulating cortical afferents were both significantly reduced in Shank3/F KO mice.
References
1 Chahrour, M and Zoghbi, HY. (2007) Neuron, PMID:17988628
2 Guy, J. et al (2001) Nature Genetics, PMID: 11242117
3 Janc, OA., and Muller, M. (2014) Frontiers in Cellular Neuroscience, PMID: 24605086
4 Kooy, R. et al (2000) Mol. Med. Today
5 Brickley, S. & Mody, I., (2012) Neuron
6 Fuccillo MV, Front Neurosci. 2016 Feb 9; 10:27. PMID: 26903795
7 Zhou Y et al., Neuron. 2016 Jan 6;89(1):147-62. PMID: 26687841
8 Peça J et al., Nature. 2011 Apr 28; 472(7344):437-42. PMID: 2142316
.