PsychoGenics’ immunohistochemistry (IHC) and histology services offer exceptional data quality, enabling the exploration of standard and novel biomarkers. Our high-throughput quantitative histology truly sets us apart in the preclinical CRO space.
We prioritize reproducibility and precision. By utilizing state-of-the-art microscopy and scanning equipment and automation techniques, we reduce potential human errors, consistently ensuring quality data and upholding the highest standards of histology and immunohistochemistry.
Samples and Sectioning
PsychoGenics has expertise in sectioning and staining all rodent tissue types with a particular specialty in CNS tissues. From advanced brain and spinal cord sectioning protocols to routine organ sectioning we are able to meet your needs. We are equipped for the three main standard embedding procedures (Cryo, Paraffin, Plastic) and the respective microtomes and cryostat for sectioning. Our team can advise you on the optimal preparation of samples for quantitative histology, which usually requires Uniform Systematic Random (USR) sampling to ensure comparability and reproducibility.
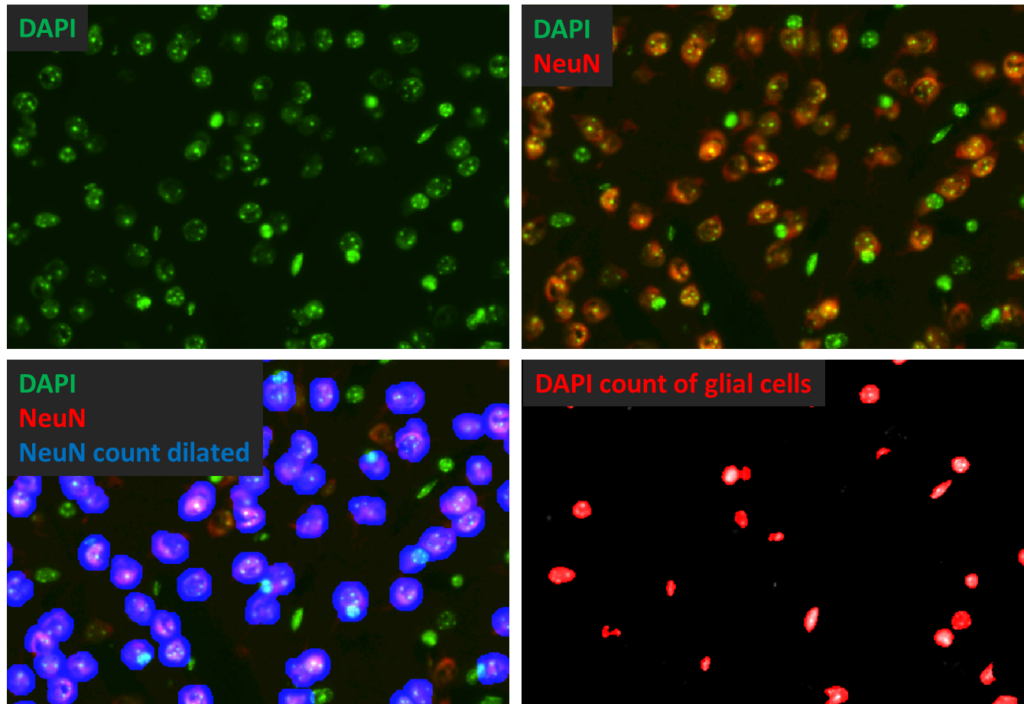
Quantitative Histology Services
Image processing and analysis have become indispensable in converting pathology into quantitative data. At PsychoGenics, we adhere to a philosophy of developing high-throughput procedures that minimize reliance on individual raters and ensure replicability at every stage of data analysis.
Our comprehensive range of histology measurements aims to provide clear and understandable results, supported by in-house macro-programming that automatically captures measurement parameters, and enables visualization and reproducibility of every count. These macros incorporate advanced techniques such as filtering, adaptive thresholding, and image arithmetic to enhance uniform object detection.
Qualitative Histology Services
A comprehensive understanding is necessary to ensure proper labeling qualification, and our team of experienced scientists and extensive consultancy network delivers. Armed with a database of validated antibody markers, we’re committed to the continuous evaluation of emerging primary antibodies, grounded in both operational and relevant biological controls.
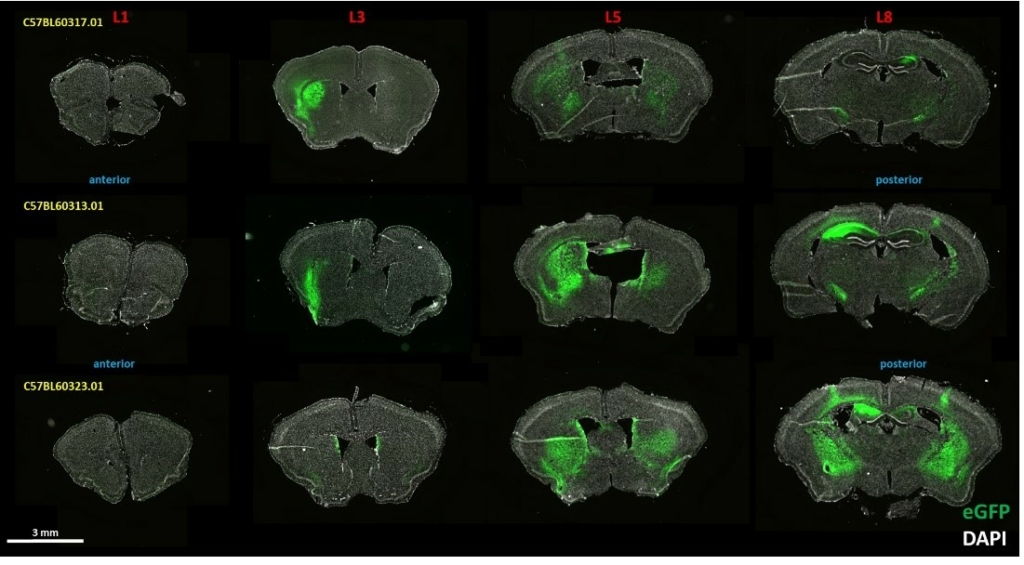
Moreover, our focus encompasses the qualitative distribution of exogenously expressed genetic material across tissues. Our team of seasoned scientists possesses expertise in various administration routes and a wide range of substances, including enhanced green fluorescent protein-labeled (eGFP) viral vectors, nanoparticles, treatment antibodies, and fluorescent dyes, as well as gene silencing reagents, such as siRNA and antisense oligonucleotides, and tagged and untagged proteins.
Immunohistochemistry Expertise
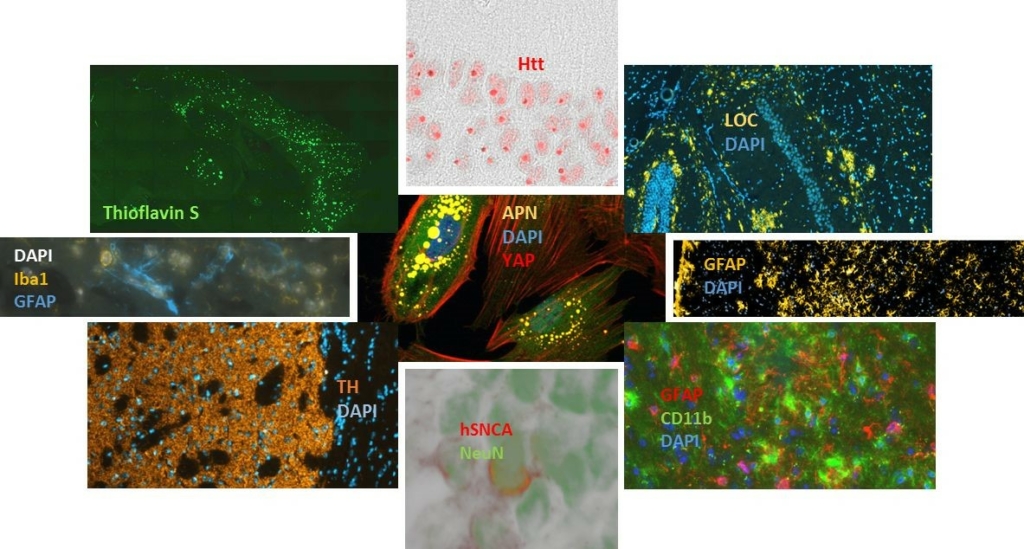
At PsychoGenics, we specialize in a comprehensive array of immunohistochemistry techniques to support your program. Our capabilities encompass both standard histochemical staining methods, as well as advanced multiple immunohistochemical labeling procedures.
We provide a diverse range of tissue staining options, including those listed below. We are continually adding new stains and antibodies (contact us if you don’t see what you need), can transfer your staining protocol, or customize a solution specifically for you.
- Nissl
- H&E
- Campbell-Switzer silver staining
- Bielschowsky silver staining
- Thioflavin S
- Congo red
- DAPI
- And many others
PsychoGenics ensures accurate and detailed visualization of tissue structures and molecular markers. Partner with us to leverage our expertise in delivering high-quality preclinical histological analyses that are tailored to your specific requirements.